About Qdenga™
Help protect your patients against dengue with Qdenga™ 1-4
Qdenga™ is supported by a robust Phase 1, 2 and 3 clinical development programme spanning 19 clinical trials, 28,175 participants, 88 study sites and 13 countries 5-21

Qdenga™ is indicated for the prevention of dengue disease in individuals from 4 years of age.1
The use of Qdenga™ should be in accordance with official recommendations.
Study Design
TIDES: Study design
TIDES: Tetravalent Immunisation against Dengue Efficacy Study in Healthy Children 2,3
- TIDES is a pivotal, ongoing, multinational, double-blind, randomised, placebo-controlled, Phase 3 study evaluating the efficacy, safety and immunogenicity of QdengaTM against dengue fever 1-3
- 20,071 healthy participants aged 4-16 years from 8 dengue-endemic countries were given at least one dose of the vaccine or placebo 1-3
- Brazil, Colombia, Dominican Republic, Nicaragua, Panama, Philippines, Sri Lanka and Thailand
- Balanced enrolment between Asia-Pacific (46.5%) and Latin America (53.5%)
- Both dengue-naïve and previously exposed individuals were included in TIDES
- At baseline, 27.7% of trial participants were seronegative for all four dengue serotypes 2,3
- Exclusion criteria included baseline febrile illness, impaired/altered immune function, hypersensitivity to any vaccine component, pregnancy/breastfeeding, previous receipt of dengue vaccine, and other protocol violations as per trial criteria 2,3
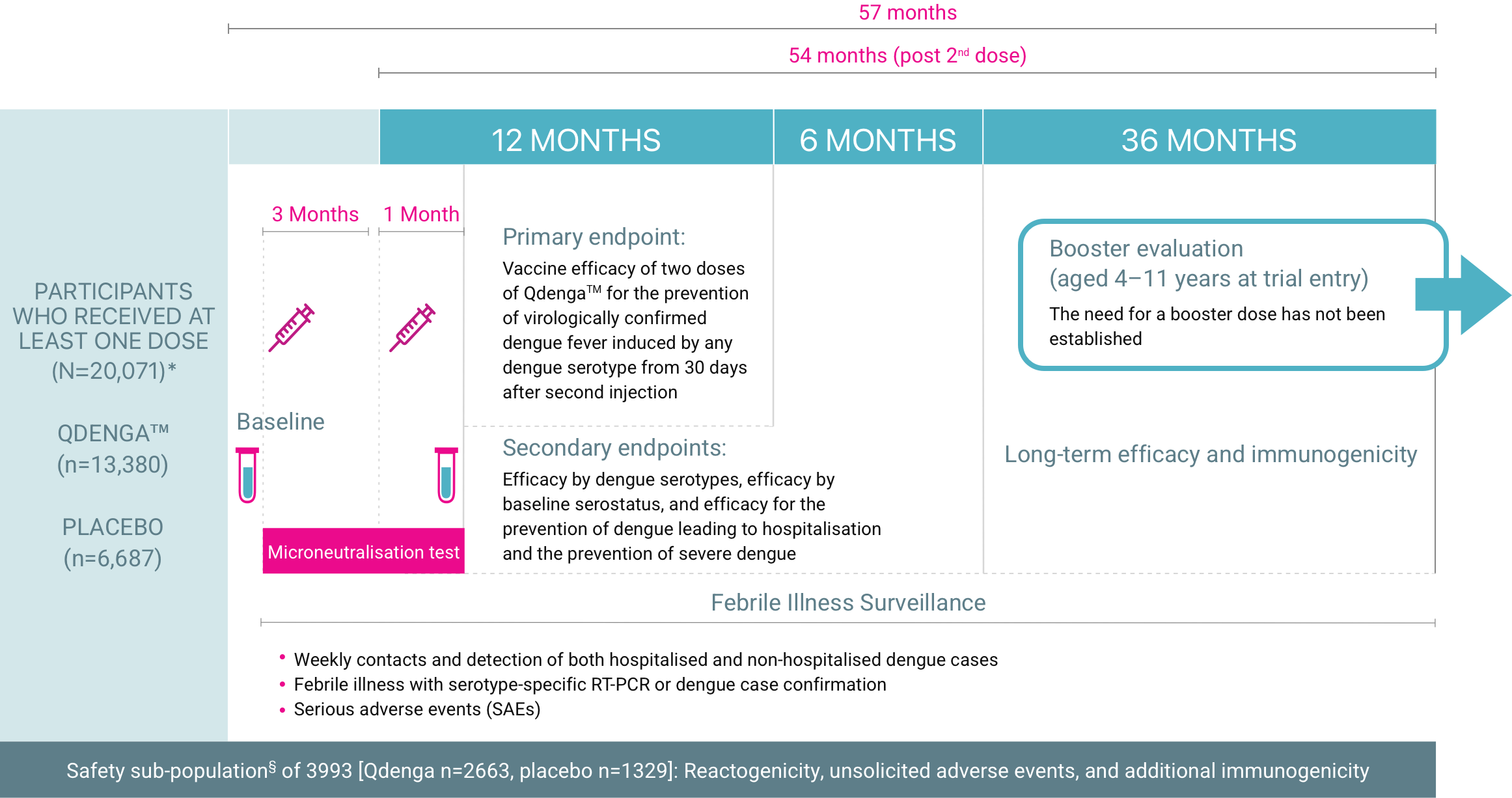

§ The safety subpopulation includes participants who were randomly selected for additional safety and immunogenicity assessments
* One 0.5 mL dose of QdengaTM contained approximately 3.6, 4.0, 4.6 and 5.1 log10 plaque-forming units of DENV-1, DENV-2, DENV-3, and DENV-4 strains, respectively; the placebo was 0.5 mL of saline. Four patients received both vaccine and placebo because of administrative errors; these participants are not included in the total.
Efficacy
TIDES: Efficacy data
TIDES: Tetravalent Immunisation against Dengue Efficacy Study in Healthy Children
The clinical efficacy of QdengaTM was assessed in TIDES, a pivotal Phase 3, double-blind, randomised, placebo-controlled study of 20,099 healthy children aged 4 to 16 years 1-3
QdengaTM effectively prevented virologically confirmed dengue (VCD) in the majority of cases 1-4
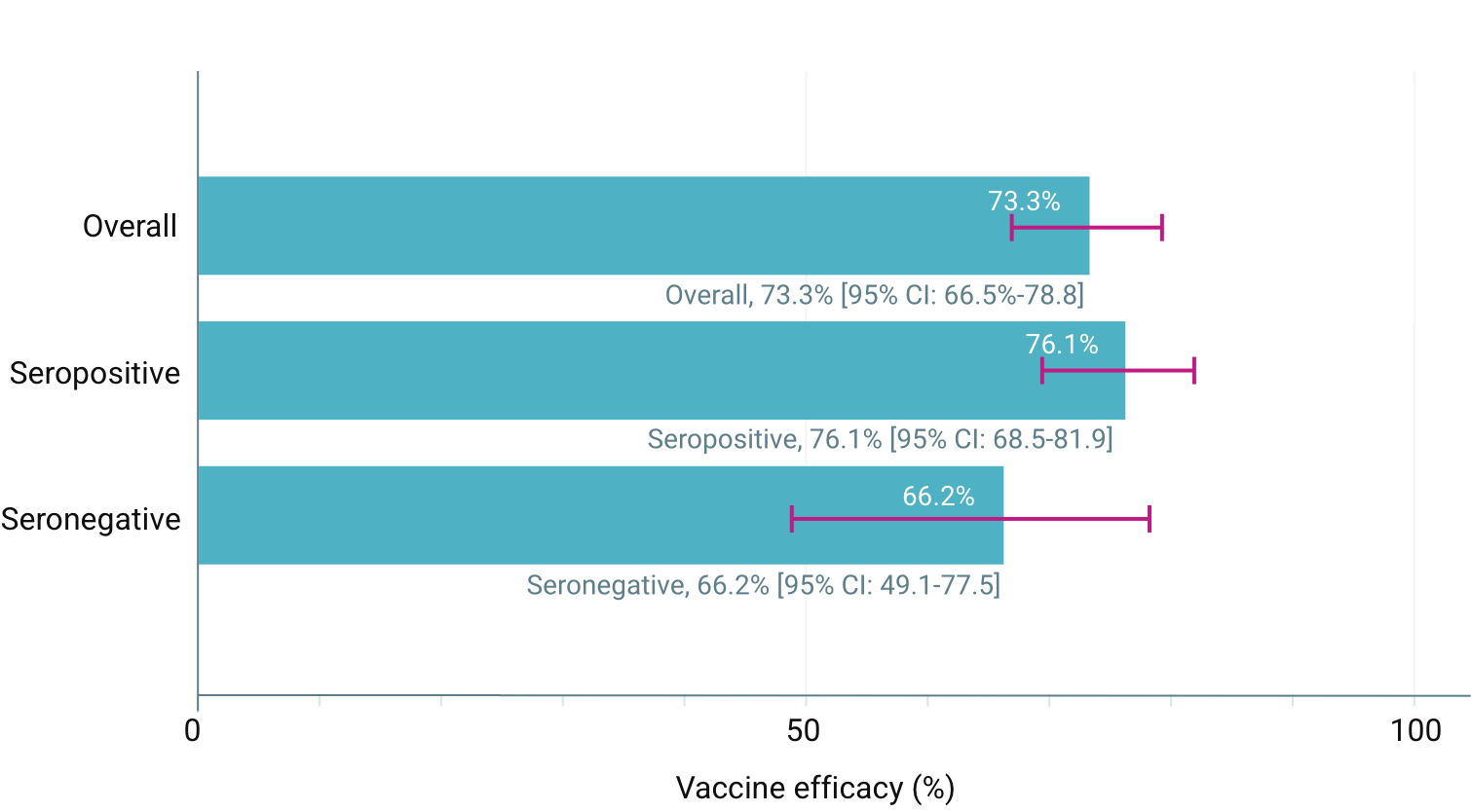
The primary endpoint in TIDES was met: QdengaTM demonstrated 80.2% overall vaccine efficacy in preventing VCD induced by any dengue serotype from 30 days to 12 months after the second dose (95% CI: 73.3%–85.3%; p<0.001) 1,2 61/12,700 (0.5%) VCD cases in the QdengaTM group vs 149/6,316 (2.4%) cases in the placebo group 1,2
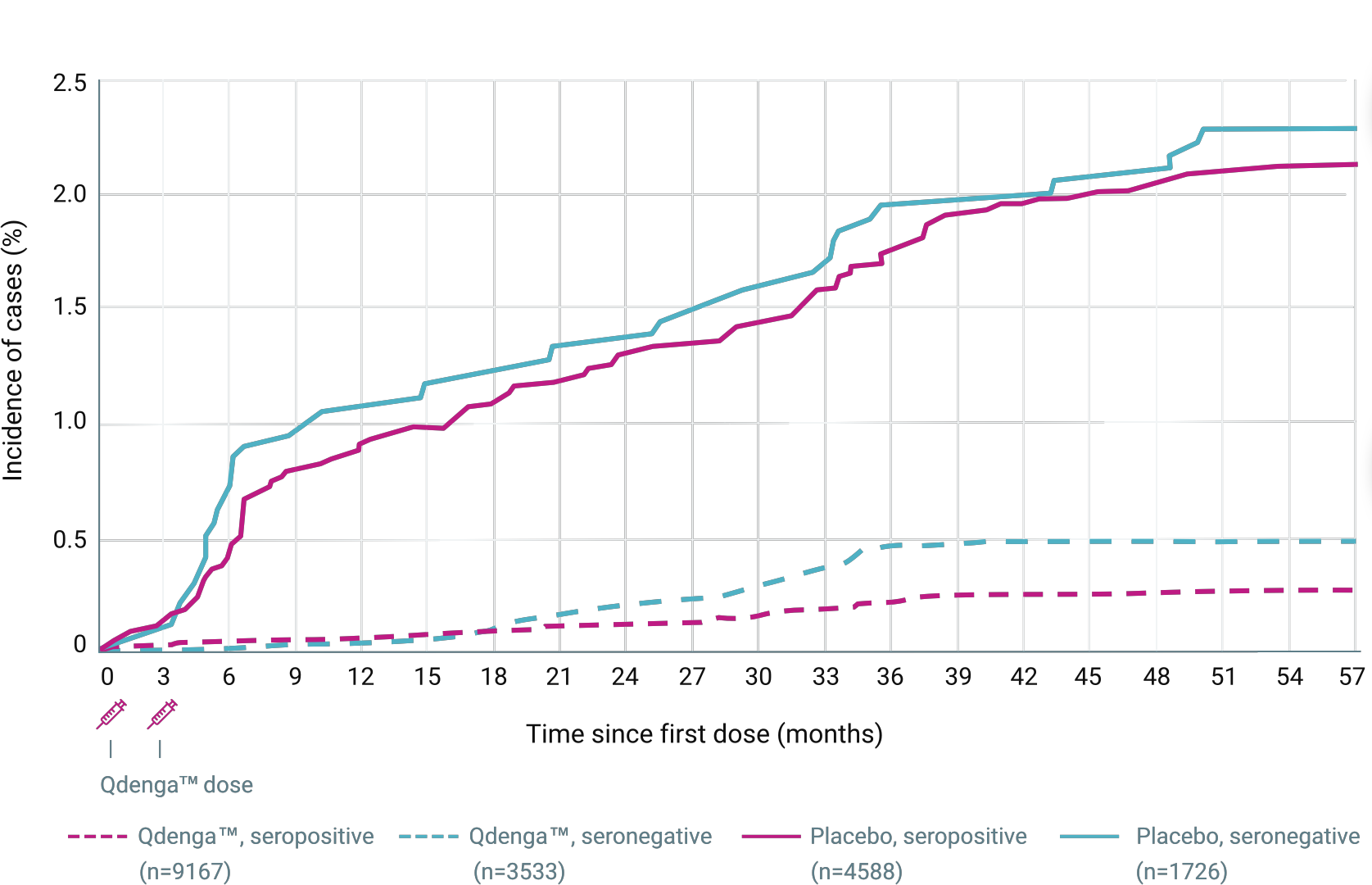
CUMULATIVE INCIDENCE OF VCD CASES* ACCORDING TO BASELINE SEROSTATUS 2-4
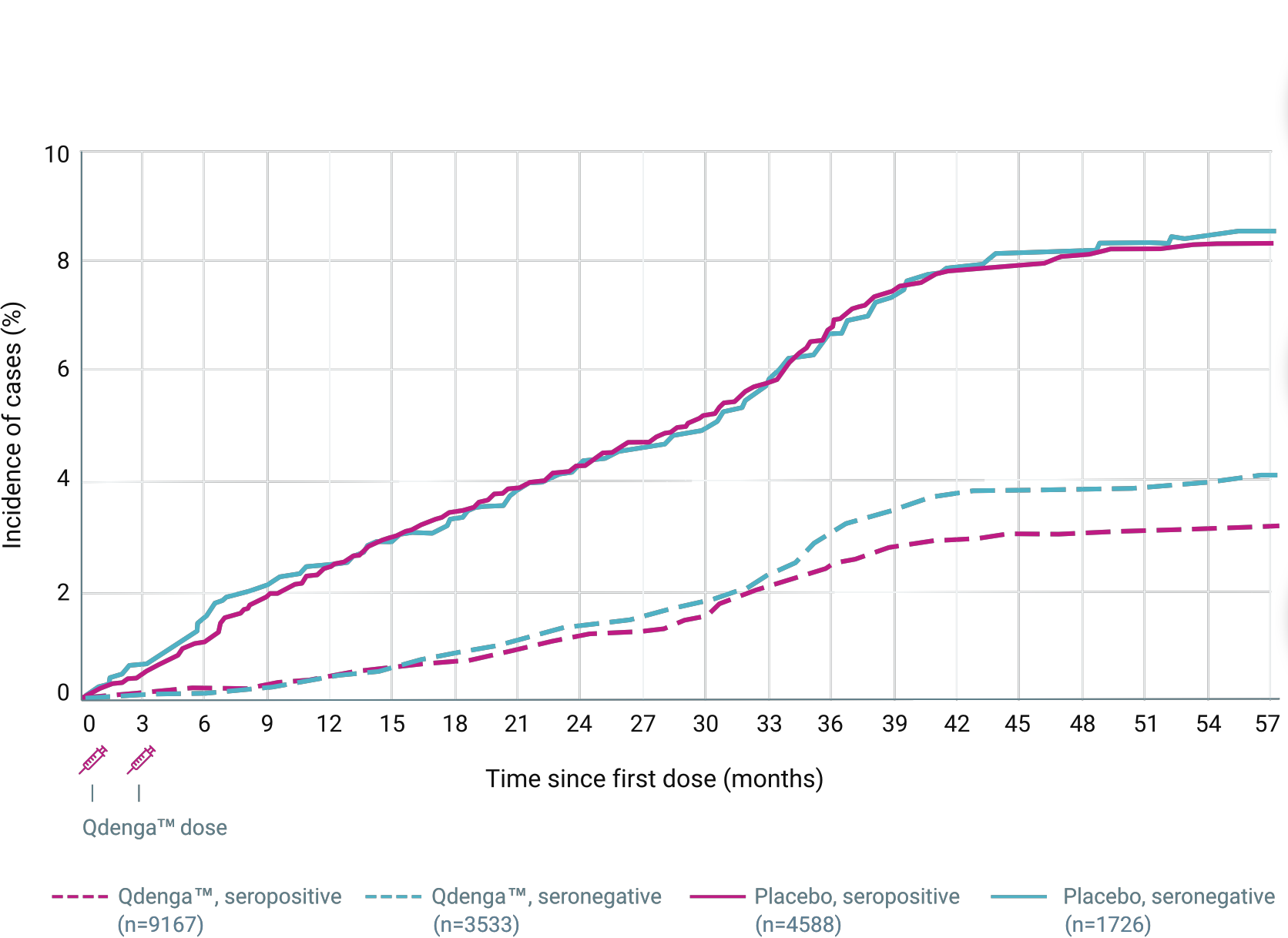
Overall vaccine efficacy 80.2% (primary endpoint) 4 to 16 years of age, 20,099 participants. From 30 days to 12 months after the second dose (95% CI: 73.3%-85.3%, p<0.001). 2 61/12,700 (0.5%) VCD cases in the QdengaTM group vs 149/6,316 (2.4%) cases in the placebo group.
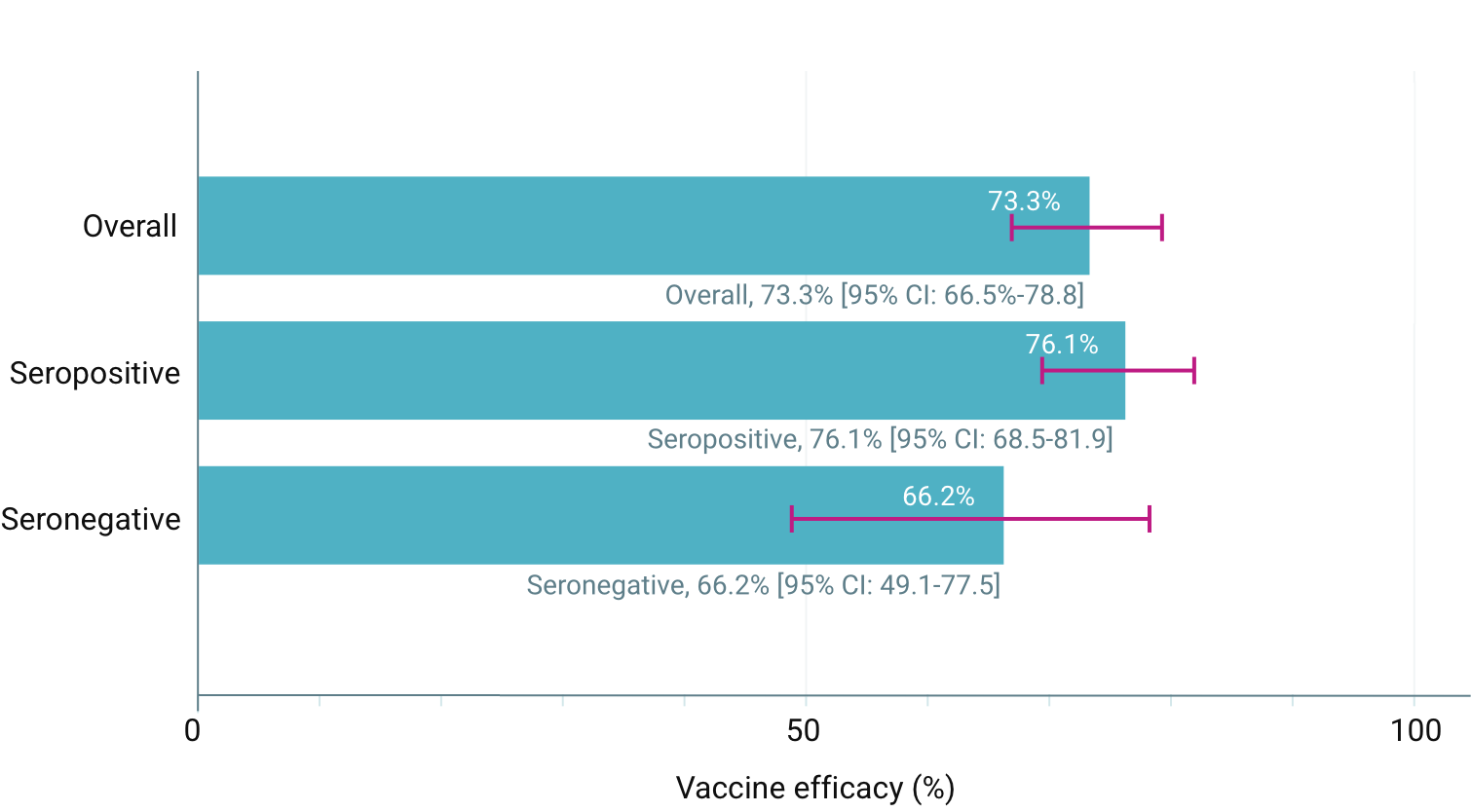
Overall vaccine efficacy 73.3% (secondary endpoint) 4 to 16 years of age, 20,099 participants. From 30 days to 18 months after the second dose (95% CI: 66.5%-78,8%). 3
114/12,700 (0.9%) dengue cases in the QdengaTM group vs 206/6,316 (3.3 %) cases in the placebo group
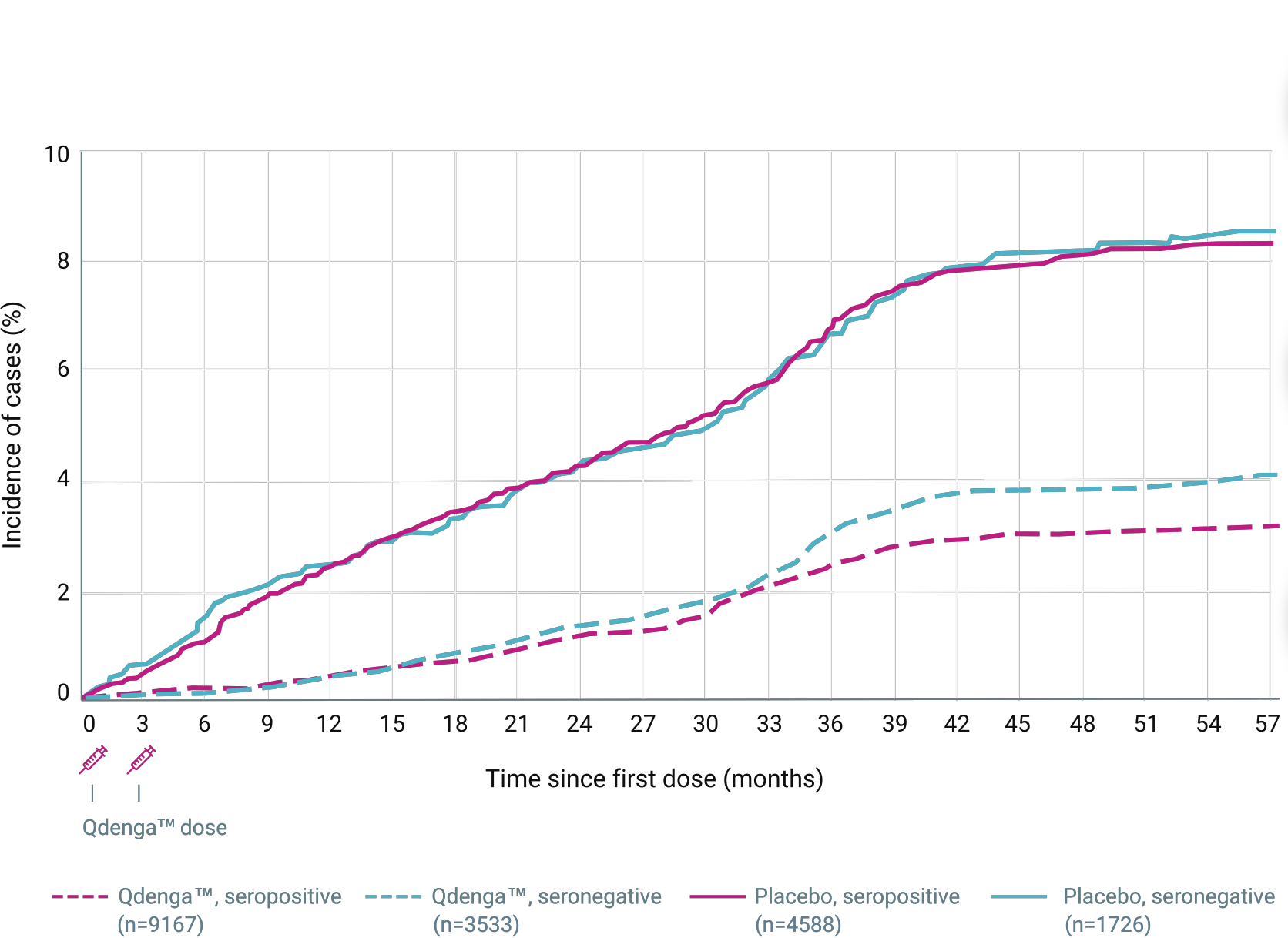
Overall vaccine efficacy 80.2% (primary endpoint) 4 to 16 years of age, 20,099 participants. From 30 days to 12 months after the second dose (95% CI: 73.3%-85.3%, p<0.001). 2 61/12,700 (0.5%) VCD cases in the QdengaTM group vs 149/6,316 (2.4%) cases in the placebo group.
Overall vaccine efficacy 73.3% (secondary endpoint) 4 to 16 years of age, 20,099 participants. From 30 days to 18 months after the second dose (95% CI: 66.5%-78,8%). 3
114/12,700 (0.9%) dengue cases in the QdengaTM group vs 206/6,316 (3.3 %) cases in the placebo group

Exploratory analysis
Exploratory analysis demonstrated 61.2% overall vaccine efficacy from first dose to 54 months after the second dose (95% CI: 56.0%-65.8%) 1,4
442/13,380 (0.9%) dengue cases in the QdengaTM group vs 547/6,687 (2.4%) cases in the placebo group1,4
Primary and secondary endpoints were analysed in the per-protocol population; cumulative incidence and exploratory evaluations were based on safety set data1
QdengaTM effectively reduced hospitalised dengue due to VCD fever in the majority of cases 1-4
The key secondary endpoint in TIDES was met: QdengaTM demonstrated 90.4% overall vaccine efficacy in reducing hospitalisation due to VCD fever caused by any dengue serotype from 30 days to 18 months after the second dose (95% CI: 82.6%-94.7%, p<0.001) 1,3
13/12,700 (0.1%) cases in the QdengaTM group vs 66/6,316 (1.0%) cases in the placebo group 1,3
CUMULATIVE INCIDENCE OF HOSPITALISED VCD CASES* ACCORDING TO BASELINE SEROSTATUS IN TIDES 2,4
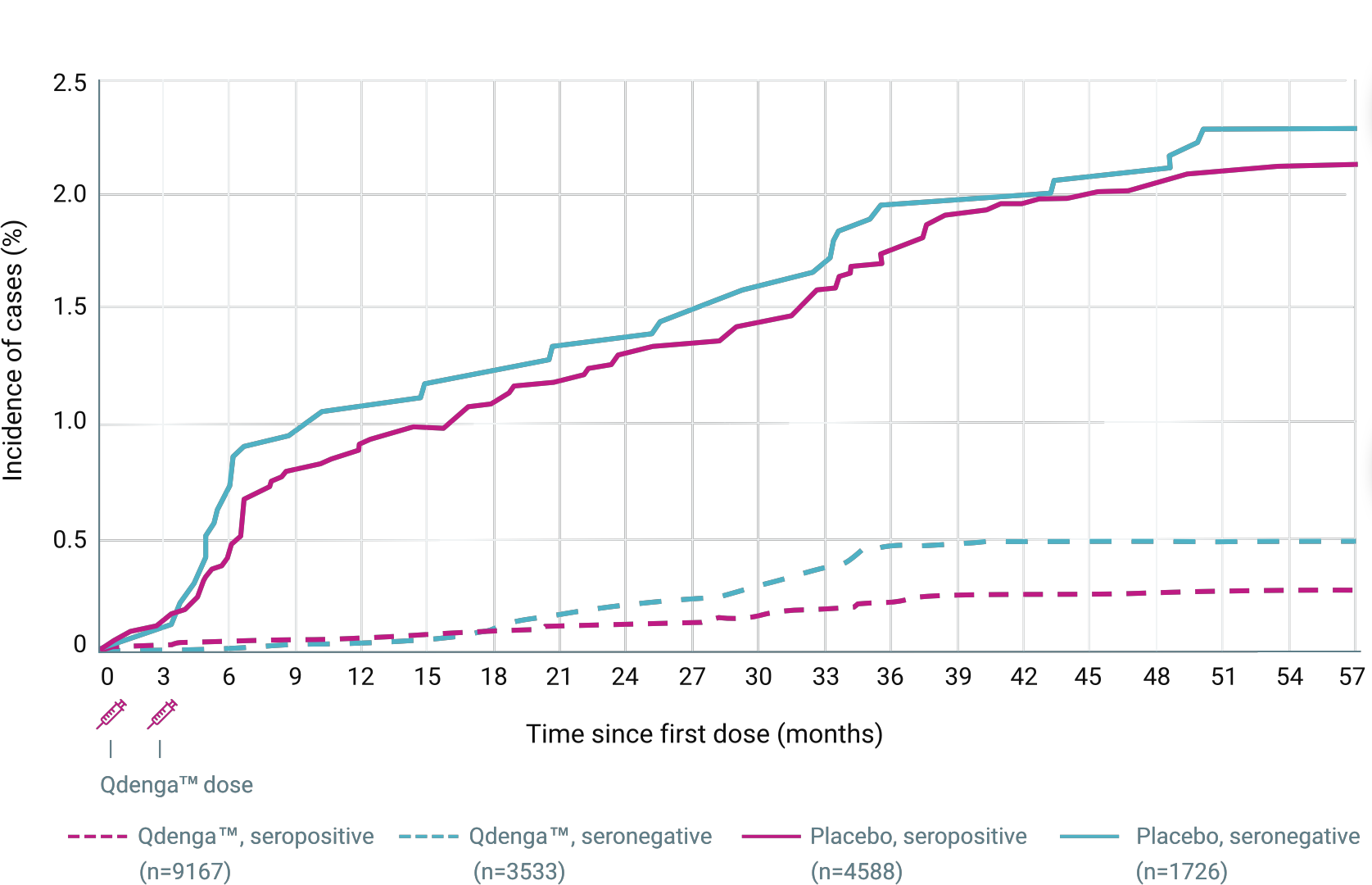
90.4% (secondary endpoint) 4 to 16 years of age, 20,099 participants. Overall vaccine efficacy in reducing hospitalisation due to VCD fever caused by any dengue serotype from 30 days to 18 months after the second dose (95% CI: 82.6% to 94.7%; p<0.001)
13/12,700 (0.1%) hospitalised cases in the QdengaTM group vs 66/6,316 (1.0%) cases in the placebo group 1,3
90.4% (secondary endpoint) 4 to 16 years of age, 20,099 participants. Overall vaccine efficacy in reducing hospitalisation due to VCD fever caused by any dengue serotype from 30 days to 18 months after the second dose (95% CI: 82.6% to 94.7%; p<0.001)
13/12,700 (0.1%) hospitalised cases in the QdengaTM group vs 66/6,316 (1.0%) cases in the placebo group 1,3

Exploratory analysis demonstrated 84.1% overall vaccine efficacy in reducing hospitalisation due to VCD fever caused by any dengue serotype from first dose to 54 months after the second dose (95% CI: 77.8%-88.6%) 1,4
46/13,380 dengue cases in the QdengaTM group vs 142/6,687 in the placebo group1
Primary and secondary endpoints were analysed in the per-protocol population; cumulative incidence and exploratory evaluations were based on safety set data1
*Specific criteria for hospitalisation were not defined in the study protocol; participants were hospitalised according to the judgement of individual investigators
90.1% reduced hospitalisations in Latin America (95% CI: 15.3%–98.8%)
1/6,806 (<0.1%) hospitalised cases in the Qdenga™ group vs 5/3,374 (0.1%) cases in the placebo group from 30 days to 18 months after the second dose 3
90.4% reduced hospitalisations in Asia Pacific (95% CI: 82.2%–94.8%)
12/5,894 (0.2%) hospitalised cases in the QdengaTM group vs 61/2,942 (2.1%) cases in the placebo group from 30 days to 18 months after the second dose 3
Efficacy of QdengaTM by serostatus, serotype and severity
EFFICACY OF QDENGATM IN PREVENTING VCD FEVER FROM 30 DAYS TO 18 MONTHS AFTER THE SECOND DOSE (SECONDARY ENDPOINTS) 1,3
EFFICACY OF QDENGATM IN PREVENTING VCD FEVER FROM 30 DAYS TO 18 MONTHS AFTER THE SECOND DOSE (SECONDARY ENDPOINTS) 3
Severity
Severity3
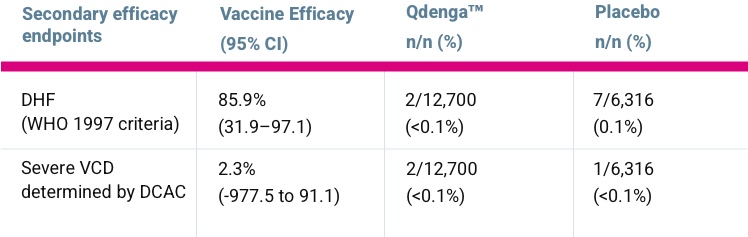
Severity
Severity3
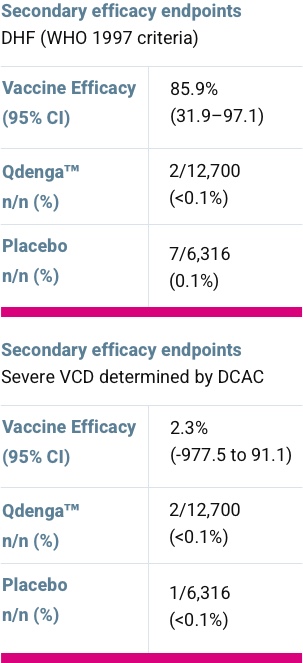

Exploratory analysis
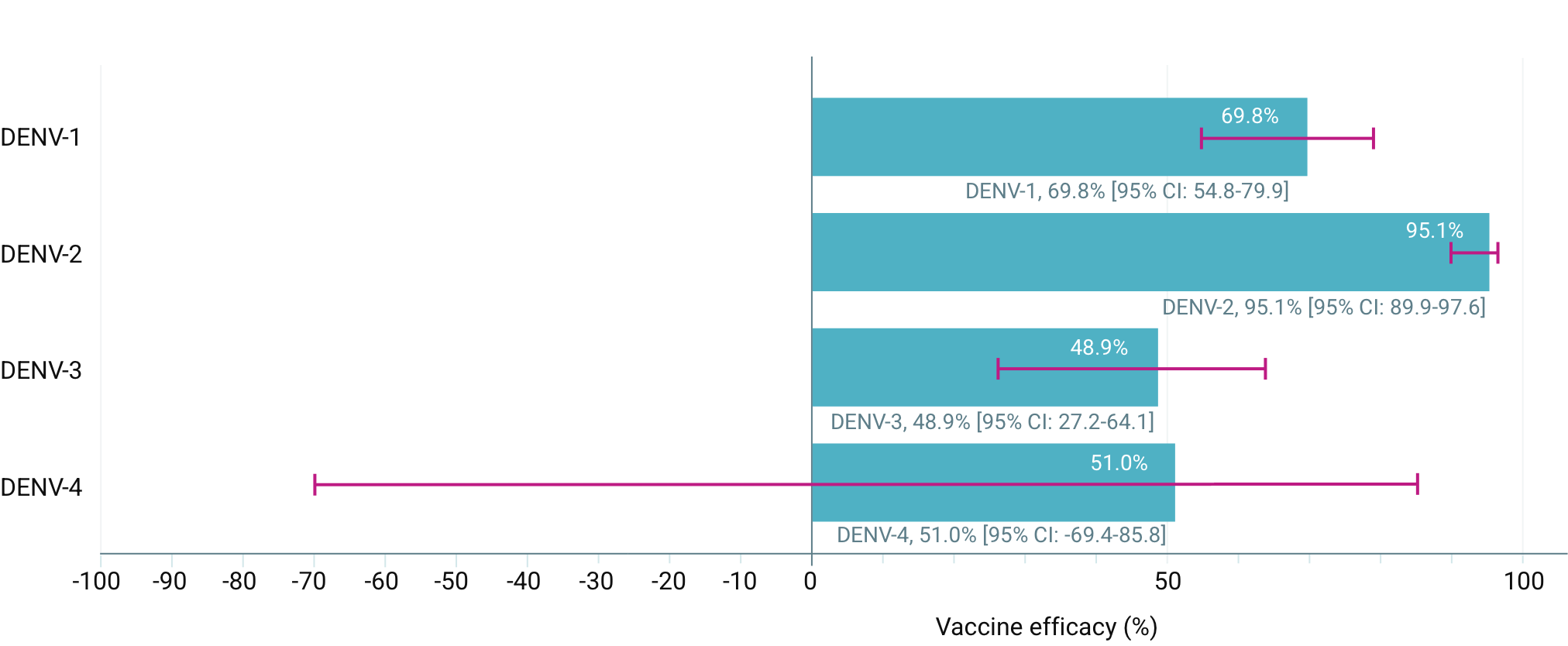
Exploratory analysis found efficacy against DENV-1 of 72.0% (seropositive, 95% CI: 52.2–83.6) and 67.8% (seronegative, 40.3–82.6), and DENV-2 of 93.7% (seropositive, CI 86.1–97.1) and 98.1% (seronegative, CI 85.8–99.7);
Analysis was not possible for DENV-4, with only one patient being seronegative at baseline. Efficacy against DENV-3 varied by baseline serostatus: 61.8% (seropositive, 95% CI: 43.0 to 74.4) and -68.2% (seronegative, 95% CI: 318.9 to 32.4); vaccine efficacy was not shown in individuals who were seronegative2
Two secondary endpoints were not met, largely due to the small number of cases: 3
- Efficacy against dengue serotype 4
- Efficacy against severe VCD (Dengue Case Adjudication Committee criteria)
Data currently suggests a lack of efficacy against DENV-3 infection in baseline seronegative participants, and there is no evidence of disease enhancement. 1,3
Efficacy of QdengaTM in people over 17 years of age
The clinical efficacy of Qdenga™ in individuals aged from 17 years of age is based on bridging of immunogenicity data 1
- Immunogenicity of QdengaTM in adults aged 18 to 60 years was assessed in DEN-304, a double-blind, randomised, placebo-controlled Phase 3 study in a non-endemic country (US) 1
- The bridging of efficacy is based on immunogenicity data and results from a non-inferiority analysis, comparing post-vaccination GMTs in the baseline dengue seronegative populations of TIDES and DEN-304 1
- Protection against dengue disease is expected in adults although the actual magnitude of efficacy relative to that observed in children and adolescents is unknown 1
GMT ratios between baseline dengue seronegative subjects in TIDES (4-16 years) and DEN-304 (18-60 years) (per-protocol set for immunogenicity) 1
Safety Profile
TIDES: Safety data
TIDES: Tetravalent Immunization against Dengue Efficacy Study in Healthy Children 2,3
Qdenga™ has been generally well-tolerated to date in TIDES 1-4
In TIDES:
- No important safety risks were identified in TIDES up to 4.5 years after the second dose of QdengaTM 4
- Similar cumulative incidences of serious AEs were observed for individuals treated with QdengaTM and placebo 3
- Up to 18 months after the second dose: 4.0% vs 4.8%, respectively
- Consistent with expected medical disorders in study population 3
- No evidence to date that QdengaTM enhanced disease severity in seronegative individuals 5
- Serious AEs occurred at 4.3% for both seropositive patients (624/14,520) and seronegative individuals (238/5,547) up to 18 months after the second dose 1,3
Overall clinical safety of QdengaTM
The overall safety profile for QdengaTM is based on a pooled analysis of 14,627 participants aged 4-60 years (13,839 children and 788 adults) who have been vaccinated with QdengaTM, including a reactogenicity subset of 3,830 participants (3,042 children and 788 adults)1
- The most frequently reported reactions in subjects 4 to 60 years of age were injection site pain (50%), headache (35%), myalgia (31%), injection site erythema (27%), malaise (24%), asthenia (20%) and fever (11%)1
- These adverse reactions usually occurred within 2 days after the injection, were mild to moderate in severity, had a short duration (1 to 3 days) and were less frequent after the second injection of QdengaTM than after the first injection1
- Frequency, type and severity of adverse reactions in children were largely consistent with those in adults1
- In clinical study DEN-205, transient vaccine viraemia was observed after vaccination with QdengaTM in 49% of study participants who were dengue-naïve and in 16% of study participants who had been infected with dengue before. Vaccine viraemia usually started in the second week after the first injection and had a mean duration of 4 days. Vaccine viraemia was associated with transient, mild to moderate symptoms, such as headache, arthralgia, myalgia and rash in some subjects. Vaccine viraemia was rarely detected after the second dose.1 Dengue diagnostic tests may be positive during vaccine viraemia and cannot be used to distinguish vaccine viraemia from wild type dengue infection.1
- Please refer to the Summary of Product Characteristics for the full safety profile, special warnings, precautions and contraindications to use of QdengaTM
Phase 3 TIDES trial demonstrated
The pivotal Phase 3 TIDES trial demonstrated:
Overall:
▼ This medicinal product is subject to additional monitoring.
GB & NI: Adverse Events should be reported. Reporting forms and information can be found at: www.mhra.gov.uk/yellowcard.
ROI: Adverse Events should be reported to the Pharmacovigilance Unit at the Health Products Regulatory Authority. Reporting forms and information can be found at: www.hpra.ie.
GB, NI and ROI: Adverse Events should also be reported to Takeda UK Ltd at: AE.GBR-IRL@takeda.com
- QdengaTM Summary of Product Characteristics
- Biswal S, et al. NEJM. 2019;381(21):2009-2019 and supplementary appendix.
- Biswal S, et al. Lancet. 2020;395:1423-1433 and supplementary appendix.
- Tricou V, et al. Efficacy and Safety of Takeda’s Tetravalent Dengue Vaccine Candidate (TAK-003) After 4.5 Years of Follow-Up. Presented at:44th ICMM World Congress on milatery medicine, 5-9 September 2022; Brussels, Belgium.Rivera, L. et al. Clin Infect Dis. 2021 Oct 4;ciab864. Doi: 10.1093/cid/ciab864. Online ahead of print
- Takeda. Lot-to-lot consistency of 3 lots of tetravalent dengue vaccine (TDV) in non-endemic country(ies) for dengue. ClinicalTrials.gov Identifier: NCT03423173. Updated October 20, 2020. Last accessed August, 2025. https://clinicaltrials.gov/ct2/show/study/NCT03423173
- Takeda. Immunogenicity and safety of tetravalent dengue vaccine (TDV) at the end of shelf life in healthy adults. ClinicalTrials.gov Identifier: NCT03771963. Updated June 7, 2021. Last accessed August, 2025. https://clinicaltrials.gov/ct2/show/NCT03771963
- Takeda. Immunogenicity and safety of dengue tetravalent vaccine (TDV) and recombinant 9-valent human papillomavirus vaccine (9vHPV) in participants aged ≥9 to <15 years. ClinicalTrials.gov Identifier: NCT04313244. Updated July 25, 2022. Last accessed August, 2025. https://clinicaltrials.gov/ct2/show/NCT04313244
- Takeda. Immunogenicity and safety of tetravalent dengue vaccine (TDV) co-administered with an hepatitis A virus vaccine. ClinicalTrials.gov Identifier: NCT03525119. Updated August 19, 2022. Last accessed August, 2025. https://clinicaltrials.gov/ct2/show/NCT03525119
- Takeda. Immunogenicity and safety of tetravalent dengue vaccine (TDV) in adolescents in non-endemic area(s). ClinicalTrials.gov Identifier: NCT03341637. Updated August 15, 2019. Last accessed August, 2025. https://clinicaltrials.gov/ct2/show/NCT03341637 Takeda. Immunogenicity and safety of tetravalent dengue vaccine (TDV) administered with a yellow fever vaccine in adults. ClinicalTrials.gov Identifier: NCT03342898. Updated October 8, 2020. Last accessed August, 2025. https://clinicaltrials.gov/ct2/show/NCT03342898
- Takeda. Safety and immunogenicity of Takeda's tetravalent dengue vaccine (TDV) in healthy children. ClinicalTrials.gov Identifier: NCT02948829. Updated July 25, 2022. Last accessed August, 2025. https://clinicaltrials.gov/ct2/show/NCT02948829
- Takeda. Efficacy, Safety and Immunogenicity of Takeda's Tetravalent Dengue Vaccine (TDV) in Healthy Children (TIDES). ClinicalTrials.gov Identifier. NCT02747927. Updated August 1, 2022. Last accessed August, 2025. https://clinicaltrials.gov/ct2/show/NCT02747927
- Takeda. Safety and immunogenicity with two different serotype 2 potencies of Takeda's tetravalent dengue vaccine candidate (TDV) in adults in Singapore. ClinicalTrials.gov Identifier: NCT02425098. Updated August 28, 2019. Last accessed August, 2025. https://clinicaltrials.gov/ct2/show/NCT02425098
- Takeda. Immunogenicity and safety of tetravalent dengue vaccine candidate (TDV) in flavivirus-naïve and dengue-immune adults. ClinicalTrials.gov Identifier. NCT03746015. Updated February 25, 2022. Last accessed August, 2025. https://clinicaltrials.gov/ct2/show/NCT03746015
- Takeda. Safety and immunogenicity of different schedules of Takeda's tetravalent dengue vaccine candidate (TDV) in healthy participants. ClinicalTrials.gov Identifier: NCT02302066. Updated February 24, 2020. Last accessed August, 2025. https://clinicaltrials.gov/ct2/show/NCT02302066
- Takeda. Long-term safety and antibody persistence of TDV and the impact of a booster dose. ClinicalTrials.gov Identifier: NCT03999996. Updated November 17, 2021. Last accessed August, 2025. https://clinicaltrials.gov/ct2/show/NCT03999996
- Takeda. Study to investigate the safety and immunogenicity of a tetravalent chimeric dengue vaccine in healthy volunteers between the ages of 1.5 - 45 years. ClinicalTrials.gov Identifier: NCT01511250. Updated July 18, 2019. Last accessed August, 2025. https://clinicaltrials.gov/ct2/show/NCT01511250
- Takeda. Safety and immunogenicity of three formulations of Takeda's tetravalent dengue vaccine candidate (TDV). ClinicalTrials.gov Identifier: NCT02193087. Updated July 18, 2019. Last accessed August, 2025. https://clinicaltrials.gov/ct2/show/NCT02193087
- Takeda. Impact of SC vs IM administration of DENVax (TDV) on safety and immunogenicity. ClinicalTrials.gov Identifier: NCT01728792. Updated July 18, 2019. Last accessed August, 2025. https://clinicaltrials.gov/ct2/show/NCT01728792
- Takeda. A comparison of the safety and immunogenicity of various schedules of dengue vaccine in healthy adult volunteers. ClinicalTrials.gov Identifier: NCT01542632. Updated July 18, 2019. Last accessed August, 2025. https://clinicaltrials.gov/ct2/show/NCT01542632
- Takeda. Phase 1b study investigating safety & immunogenicity of TDV given intradermally by needle or needle-free PharmaJet Injector. ClinicalTrials.gov Identifier: NCT01765426. Updated July 18, 2019. Last accessed August, 2025. https://clinicaltrials.gov/ct2/show/NCT01765426
- Takeda. Safety and immunogenicity study to assess TDV, a live attenuated tetravalent vaccine for prevention of dengue fever. ClinicalTrials.gov Identifier: NCT01224639. Updated June 19, 2018. Last accessed August, 2025. https://clinicaltrials.gov/ct2/show/NCT01224639
- Huang CY-H, et al. J Virol. 2003;77(21):11436-11447.
- Osorio JE, et al. Vaccine. 2011;29(42):7251-7260.
AE, adverse event; CI, confidence interval; DCAC, dengue case adjudication committee; DENV, dengue virus; DHF, dengue hemorrhagic fever; GMT, geometric mean titer; RT-PCR, reverse transcription polymerase chain reaction; TDV, the DENV [dengue virus] strain; VCD, virologically confirmed dengue; WHO, World Health Organization.
C-APROM/GB/DENV/0189 | Date of preparation: August 2025





